Last Updated: November 29, 2025
Introduction to the Methylmalonic Acidemias
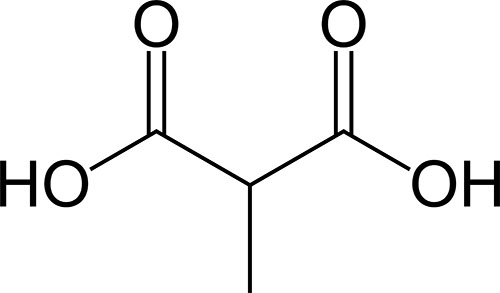
The methylmalonic acidemias represent a family of disorders that have in common the elevation of methylmalonic acid in the blood and urine. Some of the methylmalonic acidemias also include the presence of homocystinuria and are referred to as combined methylmalonic acidemia and homocystinuria.
Methylmalonic acid is an intermediate in the metabolism four amino acids, methionine, isoleucine, valine, and threonine, as well as the metabolism of fatty acids that contain an odd number of carbon atoms. The combined pathway of metabolism of these different compounds is often referred to by the mnemonic VOMIT pathway where V is valine, O is odd number carbon atom fats, M is methionine, I is isoleucine, and T is threonine. The catabolism of all five substrates yields propionyl-CoA which is converted to the TCA cycle intermediate, succinyl-CoA, by a series of three reactions catalyzed by propionyl-CoA carboxylase, methylmalonyl-CoA epimerase, and methylmalonyl-CoA mutase.
Methylmalonyl-CoA mutase is one of only two vitamin B12 requiring enzymes in humans (the other is methionine synthase). Disorders that result from defects in the genes (discussed in the following paragraphs) that are required for the proper processing and metabolism of cobalamin (vitamin B12) have historically been designated by complementation groups: cblA (MMAA gene), cblB (MMAB gene), cblC (MMACHC gene), cblD (MMADHC gene), cblE (MMTR gene), cblF (LMBRD1 gene), cblG (MTR gene), and cblJ (ABCD4 gene).
Genetics of Methylmalonic Acidemias
Inherited forms of the methylmalonic acidemias result from a number of different gene defects that occur either in the methylmalonyl-CoA mutase gene (MMUT) or in genes responsible for conversion of cobalamin (vitamin B12) to its active forms, adenosylcobalamin (AdoCbl) and methylcobalamin (MeCbl). In addition, dietary deficiency of vitamin B12 results in megaloblastic anemia accompanied by methylmalonic acidemia.
Within the family of methylmalonic acidemias there are two subfamilies, those where symptoms are associated with only elevated levels of methylmalonic acid in the blood and urine and those where the symptoms are associated with increased methylmalonic acid and increased homocystine in the blood (homocystinemia) and urine (homocystinuria). Homocystine is the disulfide bonded molecule derived from oxidation of two homocysteine molecules.
Methylmalonic Acidemias
The genetic causes of methylmalonic acidemias are due to defects in the methylmalonyl-CoA mutase gene (MMUT), the methylmalonic aciduria (cobalamin deficiency) cblA type gene (MMAA), the methylmalonic aciduria (cobalamin deficiency) cblB type gene (MMAB), and the methylmalonyl-CoA epimerase gene (MCEE).
The proteins encoded by the MMAA and MMAB genes are involved in the conversion of cobalamin to adenosylcobalamin which is required for the proper function of methylmalonyl-CoA mutase. Therefore, mutations in either of these genes will result in defective or reduced methylmalonyl-CoA mutase activity leading to methylmalonic acidemias.
Since the MCEE encoded enzyme converts D-methylmalonyl-CoA to L-methylmalonyl-CoA, defects in this gene also result in methylmalonic acidemia.
Although there are numerous genes (including those described below) that, when defective, lead to methylmalonic acidemia, over 60% of identified cases are the result of mutations in the MMUT gene that encodes methylmalonyl-CoA mutase. The organization of the MMUT and MCEE genes is described in detail in the Lipolysis and the Oxidation of Fatty Acids page as well as in the Amino Acid Catabolism page.
The MMUT gene is located on chromosome 6p12.3 and is composed of 13 exons that encode a precursor protein of 750 amino acids.
The MMAA gene is located on chromosome 4q21.31 and is composed of 8 exons that generate two alternatively spliced mRNAs, both of which encode a precursor protein of 418 amino acids. The MMAA encoded protein functions as a G-protein chaperone that delivers the product of the MMAB gene (AdoCbl) to methylmalonyl-CoA mutase. The MMAA encoded protein also protects methylmalonyl-CoA mutase from inactivation by oxidation by decreasing the rate of the formation of the oxidized inactive cofactor, hydroxocobalamin. The MMAA encoded protein also promotes the replacement of hydroxocobalamin in methylmalonyl-CoA mutase for AdoCbl, thereby restoring the activity of the enzyme.
The MMAB gene is located on chromosome 12q24.11 and is composed of 14 exons that encode a precursor protein of 250 amino acids. The MMAB encoded protein is the ATP-dependent enzyme that synthesizes AdoCbl from cobalamin [the cob(I)yrinic acid a,c-diamide form] within the mitochondria.
Methylmalonic Acidemias with Homocystinuria
The genetic causes of methylmalonic acidemias with homocystinuria are due to defects in the methylmalonic aciduria (cobalamin deficiency) cblC type, with homocystinuria gene (MMACHC), the methylmalonic aciduria and homocystinuria, cblD type gene (MMADHC), LMBR1 domain containing 1 gene (LMBRD1; cblF complementation group), the ATP binding cassette subfamily D member 4 gene (ABCD4; cblJ complementation group), and the host cell factor C1 gene (HCFC1). Mutations in the MMACHC, MMADHC, LMBRD1, ABCD4, or HCFC1 genes all affect the early steps in the overall processes of converting cobalamin to its co-enzyme forms, AdoCbl and MeCbl leading to deficiencies in both of these important cofactors.
Since MeCbl is required by methionine synthase (also called homocysteine methyltransferase) for the conversion of homocysteine to methionine, homocysteine builds up in the blood and methionine is depleted. The excess homocysteine can be oxidized to form a disulfide bond with itself forming homocystine which is excreted in urine (homocystinuria). Within this spectrum of disorders mutations in the MMACHC gene are the most common.
The MMACHC gene is located on chromosome 1p34.1 and is composed of 4 exons that generate two alternatively spliced mRNAs encoding isoform 1 (282 amino acids) and isoform 1 (225 amino acids). The MMACHC encoded protein catalyzes the reductive dealkylation of AdoCbl and MeCbl and the decyanation of cyanocobalamin (CN-Cbl).
The MMADHC gene is located on chromosome 2q23.2 and is composed of 8 exons that encode 296 amino acid mitochondrial protein. The MMADHC encoded protein controls the proportion of AdoCbl and MeCbl by promoting the oxidation of cobalamin bound to the MMACHC encoded protein.
The LMBRD1 gene is located on chromosome 6q13 and is composed of 17 exons that generate four alternatively spliced mRNAs that collectively encode proteins of 540 amino acids (isoform 1) and 467 amino acids (isoform 2). The function of the LMBRD1 encoded protein is to serve as a lysosomal transporter for cobalamin.
The ABCD4 gene is located on chromosome 14q24.3 and is composed of 20 exons that generate 23 alternatively spliced mRNAs that collectively encode 12 distinct protein isoforms. The primary function of the ABCD4 encoded protein is in peroxisomal fatty acid metabolism, however, it is suggested that it also plays a role in lysosomal release of cobalamin.
The HCFC1 gene is located on the X chromosome Xq28 and is composed of 26 exons that generate two alternatively spliced mRNAs encoding a 2080 amino acid protein (isoform 1) and a 2035 amino acid protein (isoform 2). The HCFC1 encoded protein is a transcriptional corepressor and coactivator.
Clinical Symptoms of Methylmalonic Acidemias
Although the primary clinical manifestations associated with defects in the methionine synthase gene are megaloblastic anemia with homocystinuria, there can be an associated trapping of active cobalamin as methylcobalamin (MeCbl). The trapping of MeCbl can lead to reductions in the production of adenosylcobalamin (AdoCbl) required by methylmalonyl-CoA mutase. The consequent reduction in methylmalonyl-CoA mutase activity can lead to methylmalonic acidemia.
There are two cobalamin metabolism complementation groups associated with the methionine synthase reaction, cblE and cblG. Methionine synthase is encoded by the MTR gene which is the cblG complementation group. The cblE complementation group is associated with the 5-methyltetrahydrofolate-homocysteine methyltransferase reductase (MTRR) gene. The protein encoded by the MTRR gene is required for regenerating methionine synthase to a functional state following conversion of homocysteine to methionine.
The clinical spectrum of the methylmalonic acidemias is wide and can range from a benign condition to a fatal neonatal disease. The major primary symptoms include metabolic acidosis, ketosis, hyperammonemia, hypoglycemia or hyperglycemia, and neutropenia. The major secondary complications of the methylmalonic acidemias include variable developmental delay, tubulointerstitial nephritis with progressive renal failure, acute and chronic basal ganglia involvement, disabling movement disorder with choreoathetosis, dystonia and para- or quadriparesis, pancreatitis, growth
failure, functional immune impairment, and optic nerve atrophy.
Infants with isolated methylmalonic acidemia commonly present with lethargy, failure to thrive, recurrent vomiting, dehydration, respiratory distress, and muscular hypotonia. Symptoms that are less common and usually only seen in the more severe cases are hepatomegaly and coma. As to be expected, infants harboring a complete enzyme deficiency will present earlier than any of the other gene mutation classes.
Approximately 80% of infants that are null for the MMUT gene present during the first week of life and 90% before the end of the first month of life. Around 60% of infants with mutations the MMAA or MMAB genes present with symptoms after the first month of life. Infants with MMUT, MMAA, or MMAB mutations are at high risk for metabolic stroke of the basal ganglia. These events most often occur during or following an episode of metabolic decompensation. The basal ganglia stroke will manifest with extrapyramidal (nerves controlling motor activity that descend from the cortex of the brain to the spinal cord) signs. The most characteristic extrapyramidal sign seen in methylmalonic acidemia infants is choreoathetosis [involuntary movements in a combination of chorea (irregular migrating contractions) and athetosis (twisting and writhing)].
Clinical manifestations associated with methylmalonic acidemias with homocystinurias can be quite variable as well due to the various causes. In some cases there is intrauterine growth retardation and, therefore, the clinical signs are evident at birth and include microcephaly. In infants the symptoms include failure to thrive, delayed development, difficulty feeding, pallor (abnormally pale appearance), and intellectual disability.
Neurological symptoms also manifest in patients with methylmalonic acidemia with homocystinuria. These symptoms include hypotonia and seizures. Less commonly seen, but nonetheless a significant clinical manifestation, is megaloblastic anemia. The signs and symptoms of methylmalonic acidemia with homocystinuria worsen over time and can result in life-threatening complications if not properly treated. When methylmalonic acidemia with homocystinuria does not develop until adolescence or adulthood, the most common signs and symptoms are psychiatric and cognitive in nature. In these later onset patients the most common psychiatric manifestations are changes in behavior and personality, whereas they may also experience hallucinations, delirium, and psychosis. Cognitive decline includes the loss of previously acquired mental and physical abilities that result in declines in performance in school or at work, memory problems, dementia, and speech difficulties.
Some individuals with methylmalonic acidemia with homocystinuria, whose signs and symptoms begin later, in life develop subacute combined degeneration of the spinal cord resulting in numbness and weakness in the lower limbs, and difficulty walking which can lead to frequent falls. The subacute combined degeneration of the spinal cord also occurs in chronic vitamin B12 deficiency.
The accumulation of methylmalonic acid is associated with metabolism to 2-methylcitrate, propionate, and 3-hydroxypropionate, all of which either directly or indirectly inhibit complex II (succinate dehydrogenase) of the TCA cycle and the oxidative phosphorylation pathway. The inhibition of these processes results in the neuropathogenesis typical of the methylmalonic acidemias.
In addition, in the cases of methylmalonic acidemia with homocystinuria due to mutations in the methionine synthase (MTR) gene the consequent reductions in S-adenosylmethionine (SAM or AdoMet) synthesis results in loss of efficient phospholipid methylation leading to defects in myelin sheath membrane lipid homeostasis which contributes to the neural pathology of these disorders.
Treatment of Methylmalonic Acidemias
Due to the associated hyperammonemia with the methylmalonic acidemias therapeutic intervention cannot be delayed. The initial medical treatment should include limitations in protein intake along with intravenous glucose. Treatment with drugs that includes L-carnitine, hydroxocobalamin, biotin, sodium phenylbutyrate (nitrogen scavenger to reduce serum ammonia levels), L-arginine, and N-carbamylglutamate should be instituted. Long term therapy for methylmalonic acidemia patients includes L-carnitine, vitamin B12 in responsive patients, antibiotics to reduce intestinal nitrogen producing bacteria, low protein diets, isoleucine and valine supplementation, and vitamin and mineral supplementation.
With respect to B12 supplementation, methylmalonic acidemias due to mutations in the MMACHC [methylmalonic aciduria (cobalamin deficiency) cblC type, with homocystinuria] gene are almost all responsive to B12, mutations in the MUT (methylmalonyl-CoA mutase) gene are non-responsive to B12, while the other causes of methylmalonic acidemia exhibit variable levels of responsiveness to B12 supplementation.
Although the use of specialized amino acid formulations that contain minimal to no valine, isoleucine, methionine, and threonine have become widely implemented, these types of diets can lead to iatrogenic amino acid deficiencies and poor growth outcomes in many methylmalonic acidemia patients.
Trimethylglycine (betaine) supplementation has some utility at lowering serum levels of homocysteine in methylmalonic acidemia patients. The drug Cystadane® (betaine anhydrous) has been approved as an adjunctive treatment of homocystinurias, including CBS deficiency, MTHFR deficiency, and diseases of cobalamin cofactor metabolism. The utility of oral betaine is that it activates betaine-homocysteine methyltransferase which converts homocysteine to methionine but bypasses the cobalamin-dependent methionine synthase reaction. Trimethylglycine and Cystadane are used in combinations with vitamin B6, vitamin B12, folic acid, and in some forms of methylmalonic acidemias, a low methionine diet.