Last Updated: May 26, 2026
Introduction to Dipeptidyl Peptidase Family
Dipeptidyl peptidase 4 (DPP4; also DPP-4) is a member of the large family of proteases (peptidases). There are 10 protease families that are unique to higher organisms (16 protease families can be identified in the genomes of all forms of cellular life). Within this core group of 10 protease families, a multitude of proteases evolved to yield intra- and extra-cellular processes. Serine proteases are grouped into 43 families. Protease family S9 is divided into 4 subfamilies: S9A (type prolyl oligopeptidase), S9B (DPP4), S9C (acylaminoacyl peptidase), and S9D (glutamyl endopeptidase). In humans, members of the subfamily S9B include DPP4, fibroblast activation protein-alpha (FAPα), dipeptidyl peptidase-8 (DPP8), and dipeptidyl peptidase- 9 (DPP9).
Dipeptidyl Peptidase 4
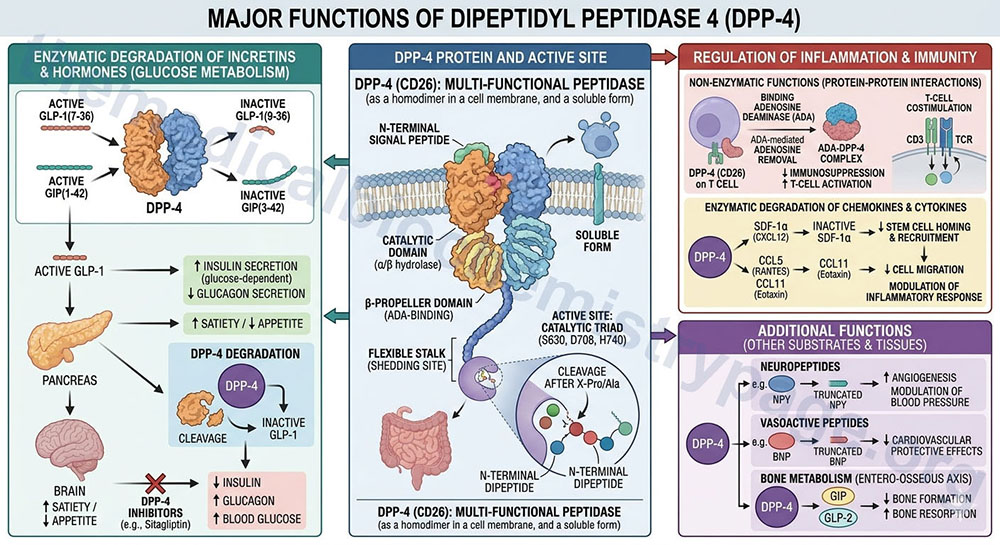
DPP4 is also known as adenosine deaminase binding protein (ADBP) or T-cell activation antigen CD26. DPP4 is a serine exopeptidase that catalyzes the release of an N-terminal dipeptide provided that the next to last residue is proline, hydroxyproline, dehydroproline or alanine. Only oligopeptides in the trans conformation are able to bind to the active site of DPP4.
The evolutionarily conserved DPP4 is found in a wide range of different organisms, including prokaryotes and eukaryotes. Comparing the structures of the prokaryotic X-prolyl dipeptidyl aminopeptidase (X-PDAP) from Lactococcus lactis and eukaryotic DPP4 demonstrates that the residues most implicated in X-PDAP activity are conserved in the same positions and orientations of DPP4, despite very different domains flanking the catalytic domain, different dimer organization, and substrate selectivity processes. Carboxypeptidases of the SC subfamily are involved in cell signaling as a consequence of their selectivity toward small peptides. Among the SC family, DPP4 is thought to be responsible for processing hormones and chemokines.
The DPP4 gene is located on chromosome 2q24.2 and is composed of 28 exons that generate four alternatively spliced mRNAs, each of which encode a distinct protein isoforms with the primary form being the longest protein of 766 amino acids. DPP4 is widely expressed in several cell types, particularly in exocrine glands and absorptive epithelia. In humans, DPP4 found in the brush borders of epithelial cells of the proximal convoluted tubules in the kidney and of the small and large intestine as well as in prostate tissue, liver hepatocytes, and activated leukocytes (T-, B- and natural killer cells).
DPP4 is a single-pass type II integral transmembrane glycoprotein which refers to the fact that its carboxy-terminus is outside the membrane and has a short N-terminal cytoplasmic extension. The DPP4 monomer is composed of a six-residue-long N-terminal cytoplasmic tail followed by a 22-residue-long transmembrane α-helix. The extracellular domain of DPP4 is divided into an eight-bladed β-propeller (Arg54-Asn497) and a C-terminal α/β-hydrolase domain (Gln508-Pro766) with the catalytic triad (Asp708, His740, and Ser630). The catalytic domain has a central eight-stranded β-sheet sandwiched by several α-helices.
Catalytic Activities of DPP4
DPP4 has a post-proline dipeptidyl amino peptidase activity, preferentially cleaving Xaa-Pro dipeptides (where Xaa refers to any amino acid) from oligopeptides. There are few Pro specific proteases and these include DPP4 and FAPα (the type II integral membrane proteins), intracellular DPPII/DPP7 that localizes to the vesicular system, soluble and cytosolic DPP8 and DPP9, and intracellular prolyl oligopeptidase (POP). The major parts of the ligand binding domain of DPP4 are defined by subdomains called S1, S2, and S3. The S1 hydrophobic pocket includes catalytic residues and is the primary determinant of substrate specificity. The hydrophobic S2 pocket consists of ionic interaction sites. In addition to its peptidase activity, DPP4 participates in adenosine deaminase binding, cell matrix adhesion, the migration and invasion of endothelial cells into collagenous matrices, co-stimulation during T cell activation, and interaction with human immunodeficiency virus (HIV) proteins.
In monomeric form of DPP4 has no enzyme activity and is only fully active as a homodimer. The regulation of DPP4 activity is observed to occur at the level of gene expression, protein synthesis and substrate selection. Interferons and retinoic acid both upregulate DPP4 gene expression through the signaling pathway that involves the signal transducer and activator of transcription alpha (STAT1α). STAT1α binds consensus sequences known as GAS motifs (interferon gamma-activated sequence), to activate CD26 gene promoters. Translation and probably translocation of DDP4 toward the cell surface can be regulated by interleukin-12 (IL-12) and tumor necrosis factor alpha (TNFα). Adenosine down regulates DPP4 by modulating mRNA expression. The down-regulation response to adenosine is not mediated through G-protein-coupled adenosine receptors, but occurs through an increase in tyrosine protein phosphatase activity resulting in a decrease in the tyrosine phosphorylation of extracellular signal-regulated kinase 1/2 (ERK1/2) mitogen activated protein kinase (MAPK) that leads to an alteration in DPP4 mRNA. DPP4 has also been implicated as a novel marker of hypoxia inducible factor-1α (HIF-1α) induction.
DPP4 Substrates
Several peptides have been identified as DPP4 substrates. These substrates include neuropeptides, chemokines, and the incretin hormones. Incretins are defined as nutrient-stimulated gastrointestinal hormones that improve oral glucose tolerance by stimulating insulin secretion. The incretins include glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP, formerly known as gastric inhibitory peptide).
GLP-1 is derived by proteolytic processing of the proglucagon protein (see the Gut-Brain Interrelationships and the Control of Feeding Behaviors page for more details). GLP-1 stimulates postprandial insulin secretion and has also been shown to stimulate proliferation and inhibit apoptosis of pancreatic β-cells. GLP-1 also inhibits glucagon release from pancreatic α-cells, inhibits gastric emptying and reduces food intake. GIP is a 42-amino acid hormone that stimulates insulin secretion in the presence of glucose. DPP4 is involved in the degradation of GLP-1, GIP, as well as glucagon to biologically inactive fragments. Thus, DPP4 inhibitors act to prevent inactivation of and prolong the duration of action of incretins, which in turn helps to correct the defective insulin and glucagon secretion that marks type 2 diabetes (see below).
The chemokines comprise a family of small (8-14 kDa) chemotactic cytokines that can be divided into sub-families on the basis of structural motifs. Their common property is leukocyte chemotaxis and, as a result, they play an essential role in modulation of immune responses. The amino terminus of chemokines interacts with the extracellular portion of chemokine receptors that are members of the G protein-coupled receptor (GPCR) family. Removal of the N-terminal amino acids of chemokines significantly alters receptor selectivity.
Peptidases, such as DPP4, are known to regulate functional activity of chemokines. Due to the expression of DPP4 by, and presentation in the plasma membrane of activated T-cells, endothelial cells, epithelial cells, and fibroblasts, chemokines have a high probability of interacting with the membrane-bound extracellular DDP4. In addition, a catalytically active soluble form of DPP4 is also present in plasma.
Chemokines that contain the N-terminal motif preferred by DPP4 could serve as substrates for the enzyme, however, data demonstrates that not all chemokines that harbor the motif are acted upon by DPP4 in vivo. This data suggests that proline in position P1 is not sufficient to result in a natural peptide being proteolyzed by DPP4. The biological functions of stromal cell-derived factors 1α and 1β (SDF-1α and SDF- 1β, CXCL12), macrophage-derived chemokine (MDC, CCL22), interferon-inducible T cell α-chemoattractant (ITAC, CXCL11), inflammatory protein-10 (IP10, CXCL10), monokine-induced by gamma interferon (MIG, CXCL9), eotaxin (CCL11), granulocyte chemotactic protein 2 (GCP2, CXCL6), and Groβ (CXCL2) have been shown to be substrates for proteolytic modification by DPP4.
Neuropeptide Y (NPY) and peptide YY (PYY) are peptide hormones of the pancreatic polypeptide (PP) family that are involved in the neuroendocrine control of feeding associated processes. The PP family of proteins bind to a family of receptors that were originally characterized as NPY receptors. There are four NPY receptors in humans and they are designated as Y1, Y2, Y4, and Y5. For more details on the activities of NPY and PYY see the Gut-Brain Interrelationships and the Control of Feeding Behaviors page. NPY and PYY are both cleaved by DPP4, DPP8, and DPP9. NPY truncated forms are especially active at the Y2 receptor, whereas lose efficacy at the Y1 receptor. Since DPP4 is found mainly in the meninges and blood vessels, whereas DPP8 and DPP9 are ubiquitously distributed in the brain, it has been hypothesized that NPY is cleaved to the truncated NPY3-36 in the periphery by soluble DPP4 or membrane-bound DPP4. It is equally possible that NPY is cleaved neuronally by DPP8/DPP9. NPY also plays a role in the neuroimmune crosstalk as evidenced by the fact that it is also produced by immune cells in response to activation and as a result modulates several immunological functions such as chemotaxis, T cell differentiation, antigen presenting cell function, leukocyte migration, neutrophil respiratory burst, and phagocytosis by binding to different Y receptor subtypes.
PYY is secreted in response to neuronal and humoral factors as well as nutrients and is a major gut-derived hormone involved in signaling satiety (the sensation of being full). Circulating PYY3-36 is derived from PYY1-36 through DPP4-mediated cleavage of the N-terminal residues. While PYY1-36 binds to all the Y receptors, PYY3-36 has a higher affinity for Y2 receptors. PYY3-36 inhibits food intake via the Y2 receptors, suppresses NPY release, inhibits gastrointestinal secretion and motor activity, and has a vasoconstrictive effect on several vascular beds. Thus, alteration in the activity of DPP4 could have pronounced effects on the responses of the brain to PYY.
Interactions with Adenosine Deaminase (ADA)
The enzyme adenosine deaminase (ADA) catalyzes the irreversible deamination of adenosine to inosine in the purine catabolic pathway. Adenosine is continuously produced both intracellularly and extracellularly. Intracellular adenosine is generated by AMP dephosphorylation or by hydrolysis of S-adenosylhomocysteine (SAH). SAH is transported into the extracellular space by specific bi-directional transporters. Extracellular hydrolysis of ATP to adenosine is catalyzed by extracellular soluble 5′-nucleotidases. Extracellular adenosine can be transported into cells as a function of a concentration gradient where it is phosphorylated to AMP by adenosine kinase or degraded to inosine by ADA. Adenosine has multiple physiological effects both within the central nervous system and on peripheral tissues that are mediated by the G-protein coupled adenosine receptors identified as A1, A2A, A2B, and A3.
In humans, various isoforms of ADA have been identified that differ in molecular weight, kinetic properties and tissue distribution. The ubiquitous zinc-binding ADA1 is the isoform whose deficiency is associated with severe combined immunodeficiency (SCID). ADA1 can form a 280kDa complex with a dimer of DPP4 or ADBP or adenosine deaminase complexing protein (ADCP). Five residues located within the cysteine-rich domain of human DPP4 are required for binding to ADA. ADA2 is the major isoform found associated with total ADA activity in human plasma and is likely secreted by activated peripheral blood monocytes. ADA can be found both in the cytosol and bound to the membrane (ecto-ADA) through two types of anchoring proteins, DPP4/CD26 or the G-protein coupled adenosine receptors. Evidence indicates that ADA co-localizing with adenosine receptors on dendritic cells (master antigen-presenting cells, APCs, of the immune system) and interacts with DPP4/CD26 that is expressed on lymphocytes. In this capacity ADA functions as a costimulatory signal that potentiates T cell activation and induces the production of the T-helper cell (Th1) pro-inflammatory cytokines.
T Cell Regulatory Activities of DPP4
DPP4/CD26 is a non-lineage-specific antigen, although it is upregulated upon T cell activation. CD26 has been suggested to regulate CD4+ T-cell maturation and migration, cytokine secretion, antibody production, and immunoglobulin isotype switching of B cells. However, the precise role of CD26 in T cells and other immune cells as well as the precise signaling pathway involved have not as yet been elucidated.
Besides binding to ADA, DPP4/CD26 interacts with several molecules involved in T cell function, such as mannose 6-phosphate/insulin-like growth factor II receptor (M6P/IGFIIR), CD45RO, and caveolin-1. Caveolin-1 is an integral membrane protein, enriched in lipid rafts, that is expressed on endothelial cells, macrophages, adipocytes, and smooth muscle cells. Interaction between CD26 and caveolin-1 results in recruitment of CARMA1 (caspase recruitment domain-containing membrane-associated guanylate kinase protein-1) to APCs. CARMA1 (also known as caspase recruitment domain 11, CARD11), is a CARD-containing membrane-associated guanylate kinase (MAGUK) family protein that is a central regulator of NF-κB (nuclear factor kappa-B) activation in lymphocytes.
CD26 dimerization is required for its DPP4 catalytic activity as well as its ability to bind CARMA1. Inhibition of DPP4 activity by synthetic inhibitors, such as those used to treat type 2 diabetes, results in suppression of T cell proliferation in vitro and a decrease in antibody production in vivo.
DPP4 Inhibitors in the Treatment of Type 2 Diabetes
The N-terminal dipeptides of the incretin hormones GLP-1 and GIP are rapidly cleaved by DPP4. Since degradation of GLP-1 results in an inactive insulinotropic peptide, strategies have been developed to prevent this rapid inactivation for use in the treatment of type 2 diabetes. These strategies involve the use of either DPP4-resistant synthetic analogues of GLP-1 (termed GLP-1 mimetics) or DPP4 inhibitors.
Using synthetic DPP4 inhibitors requires a high degree of selectivity in order to achieve an optimal safety profile. Early preclinical studies in experimental diabetes demonstrated toxicities such as anemia, thrombocytopenia, splenomegaly, lymphadenopathy, lung histiocytosis, mortality with multiple organ pathology, gastrointestinal, and central nervous system toxicity. These negative effects were most likely the result of inhibition of the closely related peptidases DPP8/9. Selective DPP4 inhibitors (second generation) were not shown to compromise immune function, as evaluated using in vitro assays of T cell activation. From these results it was concluded that the proteolytic activity DPP4/CD26 is not required for the costimulatory function.
Currently there are several selective DDP4 inhibitors approved for use in the US by the US FDA. These include sitagliptin, saxagliptin, linagliptin, and alogliptin. Vildagliptin has been approved for use by the European Medicines Agency (EMA), but has not been approved for use in the US by the US FDA.
The efficacy and safety of second generation selective DPP4 inhibitors in the treatment of type 2 diabetes has been established. The oral DPP4 inhibitors sitagliptin (marketed as Januvia®) and vildagliptin (marketed as Glavus®) have been shown to reduce the overall percentage of HbA1c by about 0.6-0.7%. Overall, these drugs are well tolerated and do not lead to severe hypoglycemia. However, all-cause infections were shown to be significantly increased after sitagliptin treatment, while did not reach statistical significance following vildagliptin therapy. Headache was often reported, especially following vildagliptin therapy. One troubling side effect of the use of DPP4 inhibitors is severe joint pain, so much so that the US FDA issued a Drug Safety Communication warning of this problem.
Although these drugs have apparent good safety profiles and have been approved for use in treating type 2 diabetes it is still important to consider the fact that the detailed activities of DPP4/CD26 have not been established. It is possible that mechanisms other than the effects on incretin activity may underlie the beneficial effects of DPP4 inhibition in type 2 diabetes. In addition, the clinical impact of DPP4 inhibitor compounds may substantially differ from that of DPP4-resistant GLP-1 mimetics. Even specific DPP4 inhibitors have potential side effects (gastro-intestinal, cardiovascular, infective, inflammatory, and allergic events) due to their prolonging the action of a number of neuropeptides and chemokines as well as to their non-enzymatic functions.