Last Updated: October 30, 2025
Introduction to Rotor Syndrome
Rotor syndrome is an autosomal recessive condition that belongs to a family of disorders that result as a consequence of defects in the metabolism and/or excretion of bilirubin. Bilirubin is the by-product of the catabolism of heme. Normal disposition of bilirubin involves its transport to the liver where it is conjugated to the sugar molecule, glucuronic acid. This conjugation reaction converts bilirubin to a water soluble compound that can be easily excreted in the feces. The conjugation of bilirubin to glucuronate is catalyzed by the enzyme bilirubin-UDP-glucuronosyltransferase (bilirubin-UGT). Rotor syndrome is related to Dubin-Johnson syndrome in that it is a disorder characterized by conjugated hyperbilirubinema.
Molecular Biology of Rotor Syndrome
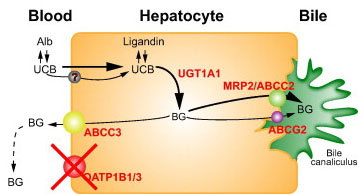
The molecular causes of Rotor syndrome are mutations in two genes. Mutations in both genes have to occur for an individual to manifest the symptoms of Rotor syndrome. These two genes encode proteins that are members of the solute carrier family of membrane transporters. The two genes are SLCO1B1 and SLCO1B3. The SCLO1B1 gene encodes the protein identified as organic anion transporting polypeptide 1B1 (OATP1B1), while the SLCO1B3 gene encodes the protein, organic anion transporting polypeptide 1B3 (OATP1B3). These two transport proteins are responsible for the uptake of bilirubin by the liver.
The SLCO1B1 gene is located on chromosome 12p12.1 and is composed of 15 exons that encode a 691 amino acid protein. Expression of the SLCO1B1 gene occurs only within hepatocytes.
The SLCO1B3 gene is located on chromosome 12p12.2 and is composed of 17 exons that generate two alternatively spliced mRNAs encoding proteins of 702 amino acids (isoform 1) and 674 amino acids (isoform 2). Expression of the SLCO1B3 gene is very nearly restricted to hepatocytes with extremely low level expression seen in testes and prostate.
Clinical Consequences of Hyperbilirubinemia
Excess circulation and accumulation of bilirubin (hyperbilirubinemia) results in a yellow-orange discoloration of the tissues and is most easily visible as icteric (yellowish) discoloration in the sclera of the eyes. Bilirubin toxicity (bilirubin encephalopathy) can be life threatening in neonates. Bilirubin encephalopathy is characterized by yellow discoloration of the basal ganglia in babies with intense jaundice and was first described over a century ago and the term “kernicterus” was coined to describe these physical changes. Any increase in plasma bilirubin above 20mg/dL is considered dangerous in neonates. However, individual differences in bilirubin sensitivity can result in kernicterus at lower bilirubin levels. Kernicterus occurs in infants with severe unconjugated hyperbilirubinemia and in young adults with high serum levels of unconjugated bilirubin, with the latter the result of inherited deficiencies in bilirubin-UGT.
Bilirubin has been shown to inhibit DNA synthesis, uncouple oxidative phosphorylation, and inhibit ATPase activity in brain mitochondria. Bilirubin also inhibits a variety of different classes of enzymes including dehydrogenases, electron transport proteins, hydrolases, and enzymes of RNA synthesis, protein synthesis and carbohydrate metabolism. All of these toxic effects of bilirubin are reversed by binding to albumin. In fact, albumin plays a vital role in the disposition of bilirubin in the body by keeping the compound in solution and transporting it from its sites of production (primarily bone marrow and spleen) to its site of excretion which is the liver.
Bilirubin levels are measured in the serum by an assay utilizing Ehrlich diazo reagent and results in the formation of an azobilirubin product. Conjugated bilirubin does not require addition of alcohol to promote the azotization reaction and thus, this is referred to as measurement of direct bilirubin. The reaction with unconjugated bilirubin requires the addition of alcohol and thus is referred to as the measurement of indirect bilirubin. Normal bilirubin measurements are 0.3–1.2md/dL for total (indirect + direct). Direct type bilirubin does not exist in the plasma, however, a small portion of indirect type bilirubin may present as direct reacting type and thus the serum measurement may show a direct bilirubin but this is never above 0.3mg/dL in a normal individual.
Clinical Features of Rotor Syndrome
Whereas, Rotor syndrome and Dubin-Johnson syndrome were originally thought to be variants of a single disorder they are now known to be different entities. Unlike Dubin-Johnson syndrome, Rotor syndrome shows no abnormal hepatic pigmentation. Although total coproporphyrin excretion in the urine of Rotor syndrome patients is markedly increased it is not to the same degree as in Dubin-Johnson patients. In Rotor syndrome homozygotes the level of coproporphyrin in the urine is around 65% and in heterozygotes it is around 40%, as compared to nearly 90% in Dubin-Johnson syndrome.