Last Updated: June 2, 2026
Introduction to Hemophilia B
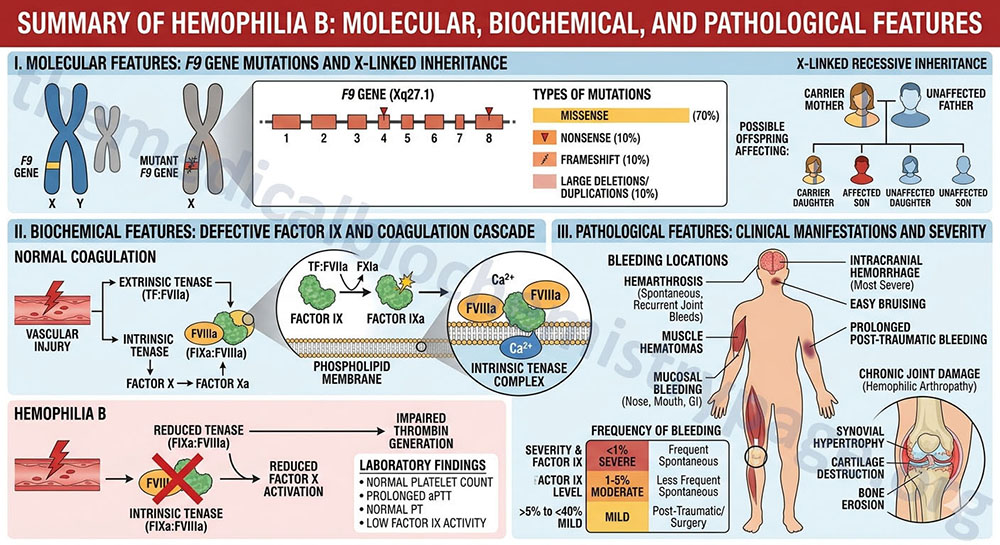
Hemophilia B, also known as factor IX deficiency or Christmas disease, is an X-linked recessive bleeding disorder caused by defects in the vitamin K-dependent enzyme, factor IX, of the clotting cascade. Factor IX is also known as the Christmas factor, hence the association of hemophilia B with the term Christmas disease. Activated Factor IX (factor IXa) is the enzyme responsible for the activation of factor X to Xa in a reaction referred to as the “tenase” complex. A cofactor in this reaction is activated factor VIII (factor VIIIa), deficiencies in which result in hemophilia A. Hemophilia B results in a bleeding disorder that is clinically indistinguishable from the more common hemophilia A. Hemophilia B occurs in approximately 1 in 30,000 male births.
Molecular Biology of Hemophilia B
Factor IX is encoded by the F9 gene. The F9 gene is located on chromosome Xq27.1 and is composed of 8 exons that generate two alternatively spliced mRNAs that encode a precursor protein of 461 amino acid (isoform 1) and a precursor protein of 423 amino acids (isoform 2).
The F9 gene resides near the FMR1 (FMRP translational regulator 1) gene that is susceptible to trinucleotide repeat expansion that is responsible for fragile X syndrome.
As of March 2023, there have been 1692 mutations identified in the F9 gene leading to hemophilia B.
Clinical Features of Hemophilia B
The major symptom of hemophilia B is spontaneous bleeding into the joints and the soft tissues of the body. The disease exhibits three levels of manifestation. The severe disease results in patients with <1% of normal factor IX protein in the circulation, moderate disease results from 1%–5% of normal factor IX protein and mild disease is found when patients have 6%–30% of normal factor IX. The most common clinical symptom of hemophilia B that requires therapeutic intervention is soft tissue hemorrhage and hemarthroses (bleeding in the joints). The leading cause of morbidity and mortality in hemophilia B patients is intracranial hemorrhage. Severely affected patients are usually diagnosed before age 1. The most effective treatment for hemophilia B patients is the administration of highly purified factor IX concentrates or, as is the more common therapeutic practice, the injection of recombinant factor IX protein.
Treatment of Hemophilia B
The treatment of patients with hemophilia B primarily involves replacing the missing factor IX clotting factor. Current therapy involves the use of recombinant human factor IX as the primary treatment protocol. The use of human factor IX derived from plasma is still being employed but is not the method of choice. The limitation of the use of recombinant humans factor IX is the short half-life in the blood (~12 hr) compared to naturally derived coagulation factors. The conjugation of recombinant factor VIII to either IgG1, albumin, or polyethylene glycol (PEG) have lead to longer half-lives of recombinant factor IX.