Last Updated: October 30, 2025
Introduction to Gilbert Syndrome
Gilbert (pronounced “zheel-BAIR”) syndrome is an autosomal recessive disorder that belongs to a family of disorders that result as a consequence of defects in the metabolism and/or excretion of bilirubin. Bilirubin is the by-product of the catabolism of heme. Normal disposition of bilirubin involves its transport to the liver where it is conjugated to the sugar molecule, glucuronic acid. This conjugation reaction converts bilirubin to a water soluble compound that can be easily excreted in the feces. The conjugation of bilirubin to glucuronate is catalyzed by the enzyme bilirubin-UDP-glucuronosyltransferase (bilirubin-UGT).
Molecular Biology of Gilbert Syndrome
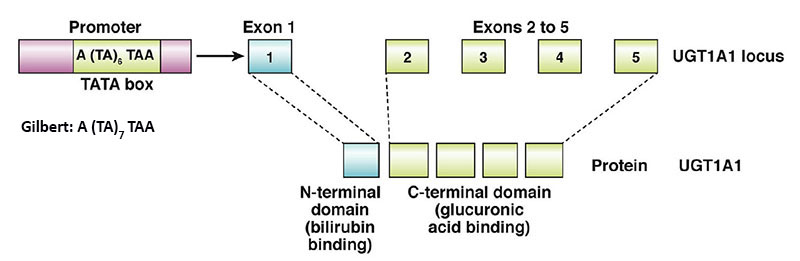
The human genome contains a complex locus on chromosome 2q37.1 that encodes several UDP-glucuronosyltransferase genes. This locus is referred to as the UDP-glucuronosyltransferase 1 family, polypeptide A complex locus (UGT1A). Several UGT1A enzymes, including bilirubin-UGT (identified as UGT1A1), are encoded by the UGT1A gene complex.
The 5′ region of the UGT1A complex contains 13 tandemly arrayed first exons, including four pseudo exons. These tandemly arrayed exons are identified as 1A1, 1A2, 1A3, etc. Exons 2, 3, 4, and 5 are located in the UGT1A 3′ region. All UGT isoforms contain the same C-terminal domain encoded by exons 2 through 5. Each first exon has its own promoter element.
The nine viable first exons are independently spliced to the common exons 2 through 5 to generate 9 UGT1A transcripts with unique 5′ ends and identical 3′ ends. The N-terminal region encoded by each unique first exon determines acceptor substrate specificity, while the 246-amino acid C-terminal region encoded by the four common exons specifies interactions with the common donor substrate, UDP-glucuronic acid.
The wild-type UGT1A1 allele is identified as UGT1A1*1. Over 135 variants/polymorphisms have been identified in the UGT1A1 allele. The bilirubin-UGT isoform (UGT1A1) consists of 533 amino acids.
The majority of patients with Gilbert syndrome inherit the disorder in an autosomal recessive manner. These individuals acquire the disorder as a result of mutations in the TATA-box of the promoter region upstream of exon 1 in the UGT1A1 gene which results in reduced levels of expression of a normal bilirubin-UGT enzyme. The normal sequence of the UGT1A TATA-box is A(TA)6TAA. The most common mutation observed in patients with Gilbert syndrome is the addition of a T-A dinucleotide into the TATA-box resulting in the sequence: A(TA)7TAA. This mutation is referred to as the UGT1A1*28 allele.
Additional TATA-box mutations, but much less common, result in A(TA)5TAA and A(TA)8TAA which are identified as the UGT1A1*36 and UGT1A1*37 alleles, respectively. The UGT1A1*36 allele is associated with increased promoter activity which is protective against hyperbilirubinemia. The UGT1A1*37 allele has promoter activity that is even less than the UGT1A1*28 allele. Both the UGT1A1*36 and UGT1A1*37 alleles are nearly exclusive to populations of African origin.
A small percentage of Gilbert syndrome individuals, particularly individuals of Asian heritage, inherited the disorder as an autosomal dominant trait. In these latter individuals the mutation in one of the UGT1A1 alleles is a missense mutation that results in reduced levels of active bilirubin-UGT since the one normal allele is not capable of producing sufficient enzyme. Both the common UGT1A1*28 homozygous UGT1A1 mutation and the rarer heterozygous missense UGT1A1 mutation found in Gilbert syndrome do not lead to the severity of reduced bilirubin-UGT activity seen in the Crigler-Najjar syndromes.
Clinical Consequences of Hyperbilirubinemia
Excess circulation and accumulation of bilirubin (hyperbilirubinemia) results in a yellow-orange discoloration of the tissues and is most easily visible as icteric (yellowish) discoloration in the sclera of the eyes. Bilirubin toxicity (bilirubin encephalopathy) can be life threatening in neonates. Bilirubin encephalopathy is characterized by yellow discoloration of the basal ganglia in babies with intense jaundice and was first described over a century ago and the term “kernicterus” was coined to describe these physical changes. Any increase in plasma bilirubin above 20mg/dL is considered dangerous in neonates. However, individual differences in bilirubin sensitivity can result in kernicterus at lower bilirubin levels. Kernicterus occurs in infants with severe unconjugated hyperbilirubinemia and in young adults with high serum levels of unconjugated bilirubin, with the latter the result of inherited deficiencies in bilirubin-UGT.
Bilirubin has been shown to inhibit DNA synthesis, uncouple oxidative phosphorylation, and inhibit ATPase activity in brain mitochondria. Bilirubin also inhibits a variety of different classes of enzymes including dehydrogenases, electron transport proteins, hydrolases, and enzymes of RNA synthesis, protein synthesis and carbohydrate metabolism. All of these toxic effects of bilirubin are reversed by binding to albumin. In fact, albumin plays a vital role in the disposition of bilirubin in the body by keeping the compound in solution and transporting it from its sites of production (primarily bone marrow and spleen) to its site of excretion which is the liver.
Bilirubin levels are measured in the serum by an assay utilizing Ehrlich diazo reagent and results in the formation of an azobilirubin product. Conjugated bilirubin does not require addition of alcohol to promote the azotization reaction and thus, this is referred to as measurement of direct bilirubin. The reaction with unconjugated bilirubin requires the addition of alcohol and thus is referred to as the measurement of indirect bilirubin. Normal bilirubin measurements are 0.3–1.2md/dL for total (indirect + direct). Direct type bilirubin does not exist in the plasma, however, a small portion of indirect type bilirubin may present as direct reacting type and thus the serum measurement may show a direct bilirubin but this is never above 0.3mg/dL in a normal individual.
Clinical Features of Gilbert Syndrome
Gilbert syndrome is also referred to as constitutional hepatic dysfunction and familial nonhemolytic jaundice. The syndrome is characterized by mild chronic, unconjugated hyperbilirubinemia. Almost all afflicted individuals have a degree of icteric discoloration in the eyes typical of jaundice. Serum bilirubin levels in Gilbert syndrome patients is usually less than 3mg/dL. Many patients manifest with fatigue and abdominal discomfort, symptoms that are ascribed to anxiety, but are not due to bilirubin metabolism. Expression of the Gilbert phenotype requires a relatively high level of bilirubin production. This is evident from the fact that persons homozygous for the UGT1A TATA-box mutation do not exhibit hyperbilirubinemia. Presentation of Gilbert syndrome symptoms also occurs more frequently in men than in women because the production of bilirubin in higher in males.